How antibiotic resistance spreads in bacterial colonies
What at first glance look like works of art (created under the influence of copious doses of psychedelic substances) are microscopy images of bacterial colonies growing on cultivation plates in the laboratory. The shimmering colours come from the fact that Yinyin Ma, Josep Ramoneda and Dave Johnson have equipped different strains of bacteria with different genes for fluorescent proteins: Some cells glow red, others green.
Mobile genetic elements
In addition, however, there is another gene for a blue fluorescent protein, which the researchers from the Department of Environmental Microbiology at Eawag have integrated into a so-called plasmid. Plasmids (in contrast to the much larger chromosomes) are small, mobile genetic elements that bacteria can exchange with each other. Plasmids may contain for example genes that neutralise the effect of antibiotics – and thus confer resistance to the otherwise lethal substances.
"Plasmids are the main vector by which antibiotic resistance is transferred between bacterial cells," says Johnson. Because the researchers added blue to the exchangeable plasmid in their experiments, they render visible what usually goes on completely unnoticed in the environment: If red bacteria receive the plasmid, they appear magenta. And green cells take on a cyan colour when they are gifted a plasmid from their neighbouring cell in magenta.
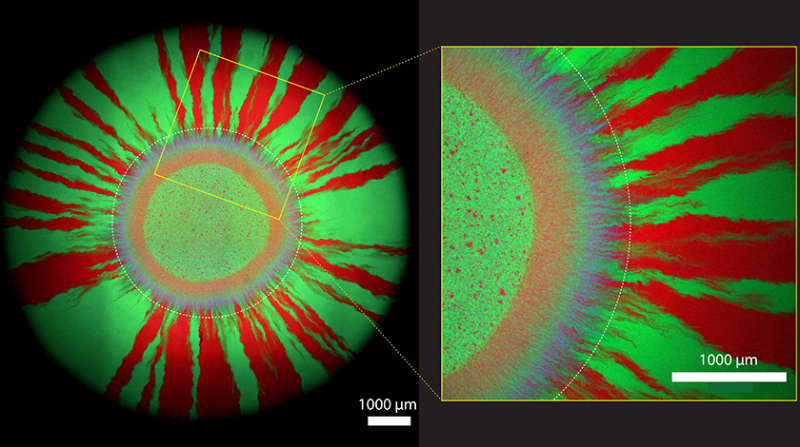
Purple wreath framed by red and green rays
Johnson's group is interested in how antibiotic resistance spreads in growing bacterial colonies. For their experiments, the researchers applied tiny drops of a mixture of green microbes and bacteria glowing in magenta to antibiotic-free culture plates. "Bacterial colonies primarily grow at the outermost edge as they expand further and further outwards," says Johnson. As shown in their microscopy images, which the researchers took a week after setting up their bacterial cultures, a purple ring (consisting of cells glowing in magenta and cyan) first forms around the tiny droplet. In this first growth zone, the bacteria exchange the plasmid most intensively with each other.
Later – i.e. further out – a pattern of green and red rays forms. Here, the cells have already lost the plasmid. This is because cells without a plasmid can divide more quickly: They save the energy that cells with the plasmid need to replicate the additional genetic element. In other words, cells without a plasmid have an evolutionary advantage – and therefore win the race when there is no antibiotic on the cultivation plate.
But on other cultivation plates, Johnson's researchers applied an antibiotic – at varying points in time. The completely different coloured patterns suggest that the plasmid is transferred to the most extent when the antibiotic is administered between 10 and 70 hours after the start of growth. Thus, the antibiotic apparently works best right at the beginning – or much later, when many cells have already lost their plasmid.
Increasing global importance
Can we conclude from these results at what point we should give antibiotics to human (or animal) patients? "No," says Johnson. That is not possible, he adds, because the experiments in the laboratory can only be compared to a very limited extent with the use of antibiotics in the clinic. There are many more than just two different strains of bacteria living on and in us humans and animals. This could be one reason why antibiotic resistances remain in the gut for years – instead of disappearing as they do on the cultivation plates.
In any case, there are still many open questions to be answered. Johnson already has a number of ideas about which aspects he would like to investigate next with his group. In this way, his group is contributing step by step to a deeper knowledge of genetic exchange between bacteria. The researchers are aware that such knowledge is of increasing global importance: "We believe that a better temporal understanding of the interplay between plasmid transfer and loss in complex microbial communities is essential to tackle the global antibiotic resistance crisis," they state at the end of their recently published manuscript.