Precisely determining toxic concentrations
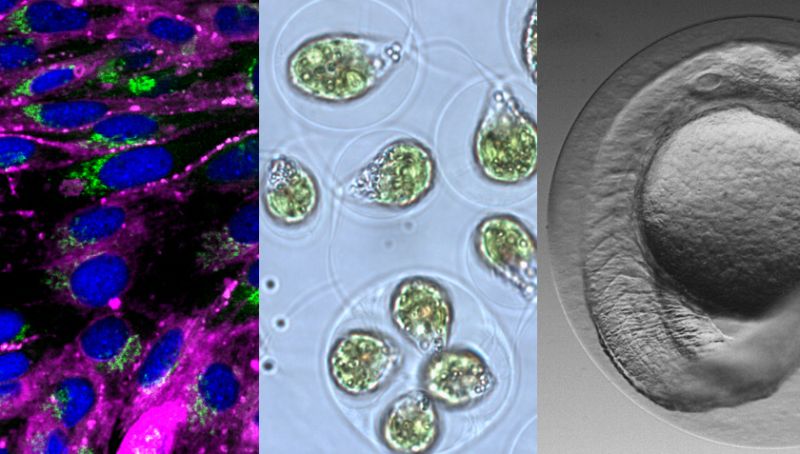
How dangerous are chemicals that enter water bodies for aquatic life? To answer this question, tests are needed on organisms – or as is increasingly the case – on isolated cells, which can replace tests on organisms. The aquatic research institute Eawag researches and develops such ecotoxicological tests. Kristin Schirmer, head of the Environmental Toxicology department and titular professor at ETH Zurich and EPFL, explains: “We want to be able to determine very precisely which effects occur at which concentrations of a chemical.” In practice, this has an influence on the ecotoxicological assessment of the chemical and can thus be incorporated into the political discussion and approval procedures.
Interpretation of the test results is difficult
Until now, however, researchers all over the world have been faced with a problem. If small volumes are used for the tests – and this is increasingly the case with modern, partly automated test procedures – the concentration of a chemical used in the sample carriers (the well plates) no longer corresponds to the concentration to which the cell or organism is exposed in the test. This can falsify the test result; in other words, the toxicity of a chemical may be underestimated.
How is this possible? “For example, there are highly volatile chemicals that evaporate during the test. Or others that bind to the wall of the well plates and are therefore less available to the cells,” Schirmer explains. Both effects can be amplified in the small volumes – due to the unfavourable volume-surface ratios coupled with the small amount of biological material. However, according to Schirmer, doing without small-scale tests would be the wrong approach, since the small amounts of chemical and the possibility of investigating many test variants simultaneously are efficient and save resources.
Therefore, researchers have so far made do by measuring the concentrations during the experiments. Although this led to accurate results, it also required time, material and money. “In terms of effort, this is comparable to conducting a second experiment at the same time,” Schirmer explains. The goal was thus clear: to find a way to predict the concentrations accurately without measuring.
A computer model provides a solution
A research team led by Julita Stadnicka-Michalak, a former scientist in Schirmer’s group, has now developed an elegant solution to this problem. A computer model enables the researchers to predict exactly which concentrations will actually affect the cells depending on the chemical and well plates used. This makes the tests far more attractive and informative – without having to carry out time-consuming chemical measurements. The Eawag model was presented to the public for the first time at the end of February in the scientific journal Scientific Reports from the renowned Nature publishing house.
Schirmer is enthusiastic about the new model: “Unlike other existing theoretical models, it is more precise and easier to apply. You do not have to know and incorporate as many parameters as with earlier models. That’s really phenomenal – and better than I had expected.”
Wide field of application
The success is based on empirical data that researchers have consistently measured and documented in all experiments over the past years. They then used these extensive data series to develop the computer model, which is the first of its kind. The model was validated by means of test series with which the researchers were able to show that the computationally predicted concentration in the well plate actually corresponds to the effectively measured concentration.
While Schirmer had expected the model to only work well with fish cells, she herself was surprised at not only how well and easily it could be transferred to other cell types such as algae, or the zebrafish embryo as a test organism, but also to numerous chemicals and different formats of the well plates. The team will now use the model in all tests and continue to assess and develop it. Schirmer is convinced that there is also a great interest among other research groups and the field of application is not limited to basic research. “As part of our spin-off Aquatox-Solutions, for example, we plan to transfer the model to industrial applications soon.”